Capsule filling machine handbook kicks off with a complete overview of the tools’s goal, perform, and numerous sorts obtainable, together with semi-automatic and computerized machines. This opening paragraph units the stage for an in depth dialogue on the machine’s elements, operations, security options, and regulatory compliance. With a short historical past of capsule filling machine improvement, we delve into the intricacies of the tools, making certain readers grasp its significance within the manufacturing course of.
The capsule filling machine handbook is an important information for operators, upkeep personnel, and high quality management groups to know the machine’s performance, guarantee its correct upkeep, and cling to regulatory requirements. By following this handbook, readers will achieve a strong understanding of the machine’s elements, operations, and troubleshooting procedures, enabling them to optimize manufacturing effectivity and product high quality.
Introduction to Capsule Filling Machines
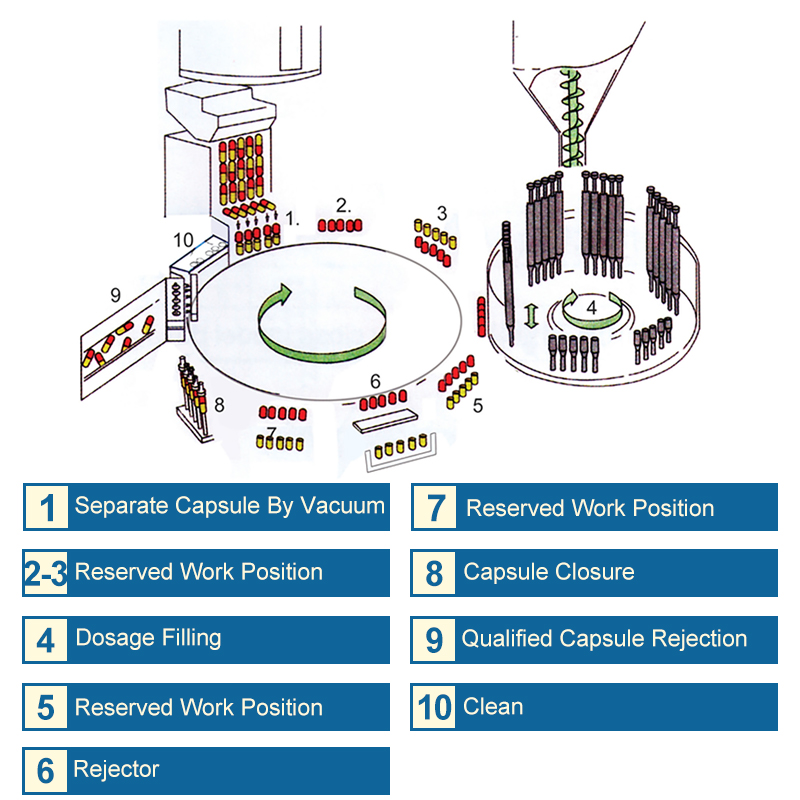
Capsule filling machines play an important position within the pharmaceutical business by automating the method of filling capsules with powdered or liquid remedy. These machines are designed to satisfy the growing demand for precision, velocity, and effectivity in pharmaceutical manufacturing.
Goal and Operate of a Capsule Filling Machine
A capsule filling machine is a tool that robotically fills capsules with the specified quantity of powdered or liquid remedy. Its major perform is to make sure correct and constant dosing, whereas additionally minimizing waste and optimizing manufacturing time. The machine sometimes consists of a hopper for storing the remedy, a filling mechanism for precisely measuring the dosing, and an ejection system for disposing of empty capsules or partially stuffed capsules.
Sorts of Capsule Filling Machines
There are two primary sorts of capsule filling machines obtainable: semi-automatic and computerized.
Semi-Computerized Capsule Filling Machines
Semi-automatic machines require handbook intervention at numerous phases of the filling course of. They are perfect for small-scale manufacturing, pilot productions, or for pharmaceutical firms that require flexibility of their manufacturing settings.
Computerized Capsule Filling Machines
Computerized machines are designed for high-speed manufacturing and are sometimes utilized in large-scale pharmaceutical manufacturing. They provide improved precision, elevated effectivity, and diminished labor prices.
Transient Historical past of Capsule Filling Machine Growth
The primary capsule filling machines have been launched within the mid-Twentieth century, with early machines consisting of handbook, labor-intensive processes. Through the years, developments in know-how have led to the event of semi-automatic and computerized machines, which have improved the precision, velocity, and effectivity of capsule filling processes.
Early Growth (Nineteen Forties-Nineteen Sixties)
The primary capsule filling machines have been handbook machines that relied on employees to fill capsules by hand. These machines lacked precision and have been time-consuming.
Mechanical Developments (Seventies-Nineties)
The introduction of mechanical filling machines marked a big enchancment within the precision and velocity of capsule filling. These machines may fill capsules in a matter of seconds, however nonetheless required handbook intervention at numerous phases.
Computerization and Automation (2000s-Current)
The appearance of computerization and automation has led to the event of high-speed, exact, and environment friendly capsule filling machines. These machines can fill capsules at charges of as much as 100 capsules per minute and provide improved accuracy by way of superior sensors and management methods.
Machine Elements and Operations
The capsule filling machine consists of a number of key elements that work collectively to fill, seal, and eject capsules. Understanding the performance of those elements is essential for working the machine effectively and successfully.
Main Machine Elements
The capsule filling machine consists of three major elements: dosers, fillers, and sealers. These elements work in conjunction to fill and seal capsules with exact quantities of powder or liquid.
The doser is liable for measuring and allotting the required quantity of powder or liquid into the capsule. It’s geared up with a precision metering system that ensures correct and constant dosing. The doser is often designed with interchangeable containers to accommodate differing kinds and portions of supplies.
The filler is liable for filling the capsule with the measured quantity of powder or liquid allotted by the doser. It’s designed to make sure exact filling accuracy, and a few fashions might embody options equivalent to computerized capping and filling peak adjustment.
The sealer is liable for sealing the capsule after it has been stuffed. It’s sometimes geared up with a heating ingredient that applies warmth to the capsule to make sure a good seal. Some fashions can also embody options equivalent to computerized ejection and capsule counting.
Filling, Sealing, and Ejecting Capsules
The capsule filling machine operates on a steady cycle, with every step rigorously synchronized to make sure environment friendly and correct processing. Here’s a transient overview of the filling, sealing, and ejecting course of:
1. Dosing: The doser measures and dispenses the required quantity of powder or liquid into the capsule.
2. Filling: The filler fills the capsule with the measured quantity of powder or liquid allotted by the doser.
3. Sealing: The sealer applies warmth to the capsule to create a good seal.
4. Ejection: The sealed capsule is ejected from the machine and picked up in a container.
Machine’s Management Panel and Person Interface
The machine’s management panel is designed to be user-friendly and intuitive, permitting operators to simply navigate and alter settings as wanted. The consumer interface sometimes features a vary of options, equivalent to:
* Show Display screen: A show display that reveals real-time details about the machine’s operation, together with filling accuracy, sealing temperature, and ejection charge.
* Buttons and Controls: A set of buttons and controls that permit operators to regulate settings and navigate the menu.
* Sensor Indicators: A set of sensor indicators that monitor the machine’s efficiency and alert operators to any potential points.
Error Messages and Options
The machine’s management panel is provided with a spread of error messages that alert operators to potential points with the machine’s operation. Listed below are some frequent error messages and their options:
* Error Message 1: “Doser failure – examine container and alter settings”.
+ Answer: Examine the doser container for injury or contamination and alter the settings to make sure correct dosing.
* Error Message 2: “Filler error – examine filling accuracy”.
+ Answer: Examine the filler’s accuracy by evaluating the stuffed capsules to a reference normal.
* Error Message 3: “Sealer malfunction – examine heating ingredient”.
+ Answer: Examine the heating ingredient for injury or malfunction and change it if obligatory.
Capsule Specs and Design

To perform effectively, a capsule filling machine requires correct and exact capsule specs. Guaranteeing correct capsule dimension consistency is essential to keep away from mechanical points and contamination dangers.
Figuring out Capsule Dimension and Materials
Capsule dimension and materials choice primarily rely upon the kind of remedy being stuffed into the capsule. There are numerous capsule sizes obtainable, starting from 0.75 ml to 0.96 ml.
* To find out the best capsule dimension, take into account the quantity of the remedy and area obtainable within the capsule.
* The commonest capsule supplies are gelatin and vegetarian capsules comprised of plant-based supplies equivalent to potato starch or rice flour.
* Gelatin-based capsules are appropriate for drugs which are heat-sensitive or simply oxidized.
Guaranteeing Capsule Dimension Consistency
Correct capsule dimension consistency is important for the machine’s efficiency and the standard of the stuffed capsules.
* Capsules of various sizes may cause the machine’s precision mechanisms to malfunction, affecting the uniformity of the stuffed capsules.
* A single misplaced capsule can stop the machine from working appropriately, resulting in delays and waste.
* Sustaining a constant provide of capsules with the identical dimension and materials will guarantee clean operations.
Creating or Sourcing Customized Capsule Designs
Generally, a particular personalized capsule design could also be required to cater to a specific remedy or buyer request.
* Capsule producers can create customized capsule designs primarily based on the supplied specs and materials necessities.
* Sourcing customized capsules from specialised producers can also be an choice, particularly for large-scale orders or quick lead occasions.
* Make sure that the chosen producer adheres to regulatory pointers and high quality requirements.
Components Affecting Capsule Weight and High quality
Capsule weight and high quality straight impression the remedy’s effectiveness and affected person compliance.
* Capsule weight impacts the dosage and stability of the remedy; an incorrect capsule dimension can result in remedy inaccuracies.
* Materials alternative influences capsule stability and compatibility with the remedy’s lively elements.
* Manufacturing processes, equivalent to temperature management, moisture ranges, and cleanliness, additionally impression the standard of the capsule.
* Capsule filling machines ought to function inside the producer’s pointers to attenuate the danger of contamination and preserve constant high quality.
Vital Dimensions and Capacities
Familiarize your self with key capsule specs and capacities:
| Dimension | Description | Normal Tolerances |
| — | — | — |
| Dimension (ml) | Quantity capability | ±5% |
| Materials | Materials thickness | ±20% |
| Size (mm) | Capsule size | ±1 mm |
| Diameter (mm) | Capsule diameter | ±0.2 mm |
| Weight Vary | Gross weight | ±5% |
Frequently examine and alter your machine settings based on the specs to make sure optimum efficiency.
Please check with the producer’s documentation for particular machine setup and calibration directions.
Machine Calibration and Troubleshooting
Common machine calibration and changes are essential to make sure optimum efficiency, accuracy, and effectivity of the capsule filling machine. Over time, the machine’s mechanisms and sensors can put on and tear, resulting in discrepancies in filling weights, capsule sealing, or machine operation. Due to this fact, periodic calibration is critical to take care of the machine’s reliability and effectiveness.
Necessity of Common Calibration
Machine calibration includes fine-tuning the machine’s parameters to make sure that it operates inside the specified vary. This course of helps to:
– Obtain exact filling weights by adjusting the dosing mechanism to account for any deviations within the machine’s efficiency.
– Optimize capsule sealing by calibrating the sealing system to make sure tight seals and reduce defects.
– Guarantee clean machine operation by adjusting parameters equivalent to motor velocity, stress, and timing to forestall vibrations, noise, or different points.
– Enhance total effectivity by lowering waste, lowering downtime, and growing manufacturing output.
Strategies for Figuring out and Fixing Widespread Technical Points
To determine and remedy frequent technical points, operators ought to observe a scientific strategy:
– Common Upkeep: Schedule common upkeep duties, equivalent to cleansing, lubricating, and changing worn-out components, to forestall machine degradation.
– Efficiency Monitoring: Constantly monitor machine efficiency, checking for any deviations in filling weights, capsule sealing, or different parameters.
– Troubleshooting Guides: Seek advice from the machine’s troubleshooting information or seek the advice of the producer’s documentation to determine potential causes and options for frequent points.
– Technical Help: Contact the producer’s technical assist staff or a certified technician for help with advanced points that can’t be resolved by way of troubleshooting.
Key Efficiency Indicators (KPIs) for Capsule Filling Machine Operation
To judge the machine’s efficiency and effectiveness, operators ought to monitor and analyze the next KPIs:
– Filling Accuracy: Measure the deviation in filling weights to make sure that they fall inside the specified vary.
– Capsule Sealing Price: Monitor the share of capsules sealed appropriately to make sure that they meet high quality requirements.
– Machine Downtime: Monitor the period of time the machine is just not in operation, together with upkeep and restore time.
– Manufacturing Output: Measure the variety of capsules stuffed per hour or per shift to judge the machine’s effectivity.
Tips for Monitoring Machine Efficiency and Effectivity
To keep up optimum machine efficiency and effectivity, operators ought to observe these pointers:
– Set Efficiency Targets: Set up and monitor efficiency targets for filling accuracy, capsule sealing charge, and manufacturing output.
– Monitor Machine Parameters: Constantly monitor machine parameters equivalent to filling weight, capsule sealing, and motor velocity.
– Analyze Information: Analyze efficiency information to determine traits, patterns, and areas for enchancment.
– Take Corrective Motion: Implement corrective motion to deal with any discrepancies in efficiency or effectivity recognized by way of information evaluation.
Regulatory Compliance and High quality Assurance

Adherence to regulatory pointers is significant within the capsule manufacturing business, making certain the manufacturing of protected and efficient merchandise. Authorities companies such because the Meals and Drug Administration (FDA) in america and the European Medicines Company (EMA) within the European Union have carried out Good Manufacturing Apply (GMP) pointers to control the manufacturing course of. These pointers are important for sustaining product high quality, making certain affected person security, and stopping contamination.
In capsule manufacturing, GMP pointers dictate the requirements for amenities, tools, personnel, and processes to make sure compliance with laws. The FDA and EMA conduct common inspections to observe adherence to those pointers. Producers who fail to adjust to laws might face extreme penalties, together with fines, product recollects, and even everlasting closure of their amenities.
On this part, we’ll focus on the significance of regulatory compliance, high quality management, and assurance in capsule manufacturing. We will even cowl the important thing documentation obligatory for regulatory compliance and strategies for making certain constant product high quality and reproducibility.
Function of High quality Management and Assurance
High quality management is an important facet of capsule manufacturing, making certain that merchandise meet the required requirements earlier than they’re launched to the market. High quality assurance, alternatively, focuses on designing and implementing processes to make sure constant product high quality. This contains setting requirements, testing, and auditing to make sure compliance with laws and inside high quality requirements.
High quality management and assurance contain a collection of steps, together with:
- Setting high quality requirements: Establishing standards for evaluating product high quality, together with specs, testing strategies, and acceptance limits.
- Testing and inspection: Finishing up common testing and inspection of uncooked supplies, in-process supplies, and completed merchandise to make sure they meet high quality requirements.
- Auditing: Conducting common audits to make sure compliance with high quality requirements, GMP pointers, and regulatory necessities.
- Corrective motion: Taking corrective motion when high quality points are detected, together with rework, rejection, or recall of affected merchandise.
- Steady enchancment: Constantly monitoring and bettering processes and methods to take care of and improve product high quality.
The position of high quality management and assurance is essential in sustaining product high quality, making certain affected person security, and stopping contamination. Producers that prioritize high quality management and assurance usually tend to obtain regulatory compliance and preserve a popularity for producing high-quality merchandise.
Key Documentation Wanted for Regulatory Compliance, Capsule filling machine handbook
Producers should preserve correct and up-to-date documentation to reveal regulatory compliance. The important thing documentation required contains:
- Certificates of Evaluation (COA): A doc that certifies the standard and purity of uncooked supplies, in-process supplies, and completed merchandise.
- Certificates of Compliance: A doc that certifies compliance with regulatory necessities and GMP pointers.
- Batch information: Information of each batch of product manufactured, together with particulars of uncooked supplies, processing circumstances, and testing outcomes.
- Validation studies: Stories that doc the validation of processes, together with tools, cleansing validation, and sanitization.
- Criticism information: Information of buyer complaints, together with investigations and actions taken to resolve the difficulty.
Sustaining correct and up-to-date documentation is important for demonstrating regulatory compliance and making certain the standard and security of merchandise.
Guaranteeing Constant Product High quality and Reproducibility
Guaranteeing constant product high quality and reproducibility requires cautious management of variables, together with uncooked supplies, processing circumstances, and tools. Producers should additionally implement high quality management and assurance measures, together with testing, inspection, and auditing to observe product high quality.
Reproducibility might be achieved by:
- Validating processes: Verifying that processes meet specified necessities, together with tools, cleansing validation, and sanitization.
- Standardizing processes: Establishing standardized processes and procedures to make sure consistency.
- Controlling uncooked supplies: Guaranteeing that uncooked supplies meet specified high quality requirements.
- Monitoring and controlling product high quality: Frequently testing and inspecting merchandise to make sure they meet high quality requirements.
Producers that prioritize constant product high quality and reproducibility usually tend to obtain regulatory compliance, preserve a popularity for producing high-quality merchandise, and forestall contamination and defects.
Final Phrase: Capsule Filling Machine Guide
In conclusion, the capsule filling machine handbook serves as a helpful useful resource for anybody concerned within the manufacturing course of. By following the rules and procedures Artikeld on this handbook, readers can guarantee protected and environment friendly operation of the tools, preserve regulatory compliance, and produce high-quality capsules. Because the business continues to evolve, it’s important to stay up-to-date with the newest developments, and this handbook gives a strong basis for understanding the advanced world of capsule filling machines.
FAQ Useful resource
Q: What are the first elements of a capsule filling machine?
A: The first elements of a capsule filling machine embody fillers, sealers, and dosers.
Q: How typically ought to I carry out upkeep on the capsule filling machine?
A: Common upkeep needs to be carried out as advisable by the producer, sometimes day by day, weekly, and month-to-month.
Q: What are the security options of a capsule filling machine?
A: Security options embody emergency cease buttons, alarm methods, and private protecting tools (PPE) necessities.